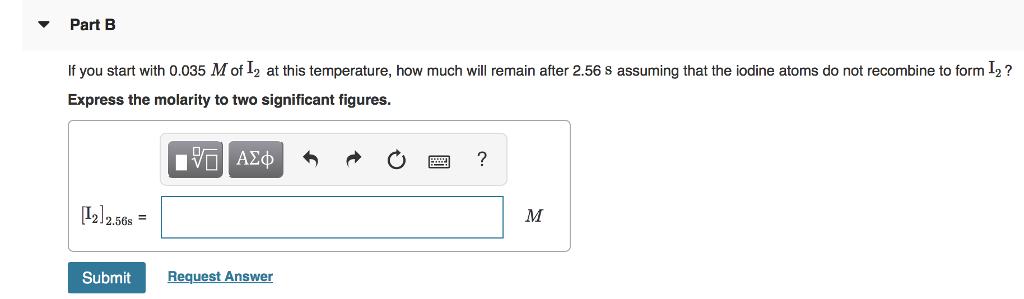 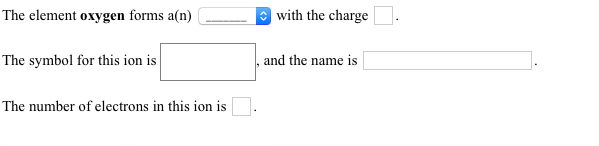
Valency is the maximum number of electrons an atom can gain or lose whereas the oxidation number is the actual number of electrons the atom gained or lost while forming a molecule. It is important to note that the valency of an atom may or may not have the same value as that of its oxidation number. Hence, we can conclude that the valency of iodine is $ - 1$. That means it has a tendency to accept an electron i.e., it will acquire a negative charge by accepting an electron. This means we need 2 iron atom, and 3 oxygen atoms, or Fe2O3. The least common multiple between 2 and 3 is 6 so we have to multiply the 3 by 2 to get 6, and the -2 by 3 to -6. Therefore, the iodine atom needs one electron to complete its octet and to reach its nearest noble gas configuration. Since the overall charge is 0, the positive and negative charges from the irons and oxygens have to sum up to 0. It is not completely soluble in water but breaks down in some organic solvents like carbon tetrachloride ($CC$Īs the number of electrons present in its valence shell $ = 7$. 
In brine pools and oceans, it is present as a water-soluble iodide on earth. Due to its larger size, it is the least reactive and most electropositive halogen. It is a dark greyish or purple blackish element which is present in the halogen family of group 17 in the periodic table. Iodine is a chemical element with atomic number $53$. Because the sum of the numbers of protons and neutrons equals the mass number, 127, the number of neutrons is 74 (127 − 53 = 74).Hint: The property of an element which determines the combining power of an atom of that element, it determines the number of other atoms with which an atom of the element can combine to form a chemical compound. The atomic number of iodine (53) tells us that a neutral iodine atom contains 53 protons in its nucleus and 53 electrons outside its nucleus. Before sharing sensitive information, make sure you’re on a federal government site. Determine the numbers of protons, neutrons, and electrons in one of these iodine anions. As an element in the production of electric light bulbs As a water purification and swimming pool sanitization chemical. Federal government websites often end in. The iodine atoms are added as anions, and each has a 1− charge and a mass number of 127. The addition of small amounts of iodine to table salt (iodized salt) has essentially eliminated this health concern in the United States, but as much as 40% of the world’s population is still at risk of iodine deficiency. Find step-by-step Chemistry solutions and your answer to the following textbook question: Use the group number to determine the charge on an ion derived from each element. 
While these are the most common valences, the real behavior of electrons is less simple. (credit a: modification of work by “Almazi”/Wikimedia Commons credit b: modification of work by Mike Mozart) Updated on NovemYou may assume that the valences of the elementsthe number of electrons with which an atom will bond or formare those that can be derived by looking at the groups (columns) of the periodic table. (b) The addition of small amounts of iodine to salt, which prevents the formation of goiters, has helped eliminate this concern in the US where salt consumption is high. Use the group number to determine the charge on an ion derived from each element. In our foods, iodine is present as an ion, iodide, in combination with another oppositely charged ion, like potassium. \): (a) Insufficient iodine in the diet can cause an enlargement of the thyroid gland called a goiter.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
